In Zaporizhzhia region, the National Police, in collaboration with representatives of the State Service of Ukraine on Medicines and Drugs Control, uncovered a scheme involving the illegal import and sale of unregistered medicines for cancer patients
Опубліковано 27.04.2026 о 13:21The Strategic Investigations Department of the National Police in Zaporizhzhia Oblast, in collaboration with investigators from the Investigative Directorate of the Main Department of the National Police in Zaporizhzhia Oblast, uncovered a scheme involving the illegal import and sale of unregistered medicines.
According to the investigation, a channel for supplying medicines from abroad in violation of established procedures was organized by businessmen in collusion with officials from a medical facility. The medicines, including those used to treat cancer patients, were imported illegally and sold to patients through pharmacies.
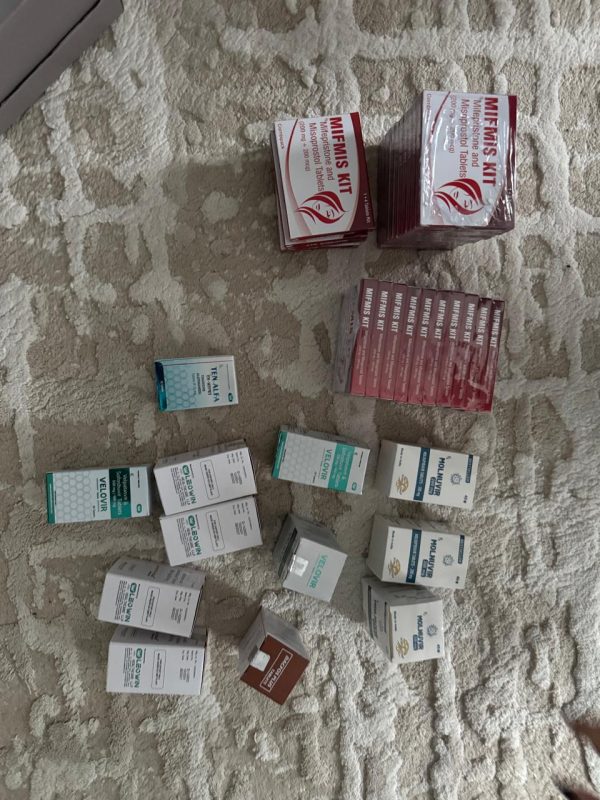
National Police officers conducted searches at the residences of individuals involved in the case, at a medical facility, a pharmacy, and the homes of other individuals linked to the scheme in Zaporizhzhia, Chernivtsi, and Chernivtsi Oblast. They seized documents, records, mobile phones, and data storage devices relevant to the investigation.
Based on the evidence gathered, charges have been filed against the pharmacy manager and the head of the hospital department. Depending on their respective roles in the scheme, their actions have been classified under Parts 1 and 3 of Article 354 of the Criminal Code of Ukraine.
The pre-trial investigation is ongoing under the procedural supervision of the Zaporizhzhia Regional Prosecutor’s Office. Further steps are being taken to hold other participants in the scheme accountable.
These measures were carried out with the involvement of specialists from the regional offices of the State Service of Ukraine on Medicines and Drugs Control in Zaporizhzhia and Chernivtsi regions.

The State Service of Ukraine on Medicines and Drug Control emphasizes to business entities to strictly comply with the requirements of the Licensing Conditions for conducting business activities related to the manufacture of medicines, wholesale and retail trade in medicines, and the import of medicines (except for active pharmaceutical ingredients), as approved by Resolution No. 929 of the Cabinet of Ministers of Ukraine dated 30 November 2016.
The State Service of Ukraine on Medicines and Drugs Control reminds consumers that if they have any doubts about the quality of purchased medicines, they can fill out an online form available on the homepage of the SMDC’s official website (dls.gov.ua) under the section “Submit a report regarding the quality/suspected counterfeiting of a medicine.”
The State Service of Ukraine on Medicines and Drugs Control has updated its advice for consumers and business entities on its official website to help them protect themselves from counterfeit medicines.
 People with visual impairment
People with visual impairment  Українською
Українською 
 Попередня
Попередня